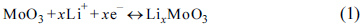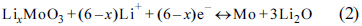3.1 Materials Characterization
The XRD patterns of the AMo-PANI, ES and AMo are shown in
Fig. 1a. The XRD pattern of ES exhibits a typical partially crystalline polymeric diffraction feature between 20 ° and 30 °, whereas AMo-PANI shows an additional diffraction peak around 7.5 ° The peaks appearing within the small diffraction angle for PANI doped with different acids have been previously observed by many researchers: for example, the diffraction peaks at 6.4 ° and 6.7 ° correspond to the doping of cardanol
[26] and 12-tungstophosphoric acid
[27], respectively. For our samples, it is believed that the peak near 7.5 ° is related to the existence of molybdate counter-ions in PANI, since the XRD pattern of ES does not show any peak around this position. It should also be noted that no diffraction peak corresponding to AMo crystal can be found in the XRD pattern of AMo-PANI, suggesting that AMo anions are homogeneously dispersed in PANI due to the electrostatic driving force towards neutrality between PANI chains and AMo anions (see
Scheme 1).
Fig. 1.
(a) XRD patterns of AMo-PANI (red), ES (blue) and AMo (black); (b) XRD patterns of Mo2C/C, MoO3/C and pristine MoO3.
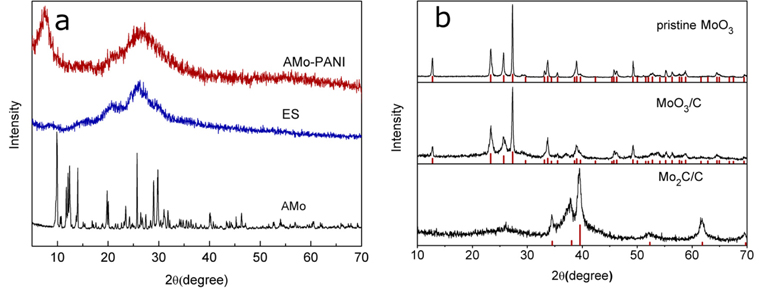
Scheme 1.
Schematic illustration of PANI doped with AMo and the formation of MoO3/C composite.
Fig. 1b shows the XRD patterns of Mo
2C/C, MoO
3/C and pristine MoO
3, respectively. The XRD pattern of Mo
2C/C is indexed to Hexagonal Mo
2C (JCPDS: 65-8766), and those of MoO
3/C and MoO
3 are indexed to orthorhombic MoO3 (JCPDS: 05-0508). A broad weak peak between 20° and 30° only emerges in the XRD patterns of Mo
2C/C and MoO
3/C which cannot be found in the pattern of pristine MoO
3. It is believed that this peak derives from the amorphous carbon pyrolyzed from the PANI chains. Moreover, it is known that AMo can be decomposed to MoO
3 around 400 ℃ at ambient pressure
[20,
23]; concomitantly, we have found that the MoO
3 and C composite can be obtained by calcining AMo-PANI at 400 ℃ under N
2 atmosphere but it portrays a poor conductive behavior (ca. 10
−5 S/cm). For enhancing the electrical conductivity of the composite, AMo-PANI was firstly calcined at a higher temperature of 800 ℃ under N
2 atmosphere to obtain Mo
2C/C (
Fig. 1b); upon oxidation of Mo
2C/C at 310 ℃ in air, a MoO
3 and C composite with excellent electrical conductivity is obtained (1.5 S/cm). Thus, it is expected that the latter composite with higher electronic conductivity is a good candidate as electrode material.
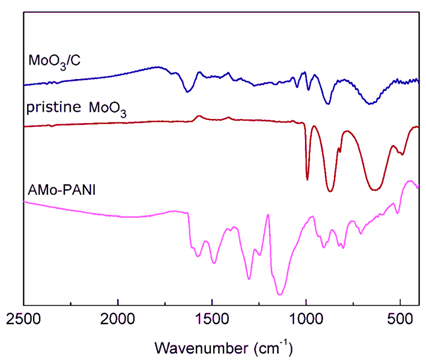
The FT-IR spectra of AMo-PANI, MoO
3/C and pristine MoO
3 are shown in
Fig. 2. typical absorption bands of PANI can be observed from the FT-IR spectrum of AMo-PANI. For example, the band located at 1143 cm
−1 is due to the quinonoid unit of doped PANI. The bands located at 1248 cm
−1 and 1302 cm
−1 are assigned to C-N stretching of the benzenoid unit, while the bands at 1494 cm
−1 and 1574 cm
−1 are assigned to C=C stretching of quinonoid and C=N stretchings of benzenoid units, respectively
[28]. The pristine MoO
3 shows three main fingerprint vibrational modes of crystalline MoO3 between 500 - 1000 cm
−1. The bands located at 648 cm
−1, 881 cm
−1 and 987 cm
−1 are attributed to the bending of Mo-O-Mo units and the vibrations of the Mo-O and terminal Mo=O moieties, respectively
[29]. In the FT-IR spectrum of MoO
3/C, the characteristic absorption bands for MoO
3 can be clearly observed. The absorption bands located at 1047 cm
−1 and 1639 cm
-−1 are attributed to C=C and C-O moieties
[30], which only appeared in the spectrum of MoO
3/C. This evidence proves that carbon is successfully incorporated within MoO
3.
Fig. 2.
FT-IR spectra of AMo-PANI (pink), pristine MoO3 (red) and MoO3/C (blue).
The FE-SEM images of MoO
3/C and pristine MoO
3 are shown in
Fig. 3. It can be observed that pristine MoO
3 presents a granular morphology (
Fig. 3a) that clearly exhibits the layered nature of MoO
3. The size of the granules lies in the sub-micrometer scale (500-1000 nm), although a few of them have a size of 20-50 nm. The SEM image shown in
Fig. 3b indicates that the MoO
3/C sample also has a similar granule morphology, their size falling in the 100-250 nm range, which is smaller than that of pristine MoO
3, evincing that the growth of granules is restricted by the amorphous carbon. The porous granules in MoO
3/C can be seen from the higher magnification FESEM image shown in the inset in
Fig. 3b, which may be related to the decomposition of PANI during the calcination process and possibly benefit their electrochemical performances. Similar micrographs of the porous structure for other TMOs and carbon nanocomposites were also previously reported
[23,
31].
Fig. 3.
(a) FE-SEM images of pristine MoO3 and (b) MoO3/C (the inset correspond to a larger magnification).

3.2 Eletrochemical Characterization
The electrochemical Li-storage properties of MoO
3/C and pristine MoO
3 were studied by CV and galvanostatic charge/discharge measurements. Two pairs of redox peaks around 1.3 V and 0.3 V for MoO
3/C and pristine MoO
3 can be observed from CV curves (
Fig. 4a). These redox peaks reflect the two stages of the lithiation/delithiation process in MoO
3 [32,
33] and the corresponding redox reactions can be described with the following reactions:
Fig. 4.
(a) CV curves, (b) cyclic performance at 0.5 A/g (initial cycle is measured at 0.1 A/g), (c) EIS plots (inset show the EIS plots of EIS with larger magnification) and (d) rate performance of MoO3/C and pristine MoO3.

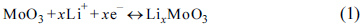
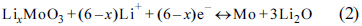
The first stage (Eqn. (1)) is assigned to the insertion of Li ions into the structure of MoO
3 to form Li
xMoO
3. The second stage (Eqn. (2)) revealing a relatively higher specific capacity than the previous stage is ascribed to the Li ions inserting into Li
xMoO
3 to form the metal of Mo and the lithium oxide. A small reduction peak near 0 V can be observed too. It seems that this peak is ascribed to the intrinsic character of MoO
3 since similar peak was observed in other literatures
[20,
29]. The MoO
3/C redox peaks are more obvious than that of pristine MoO
3. In addition, the integrated area surrounded by CV curve (i.e. charge/charge capacity) of MoO
3/C is much larger than that of pristine MoO
3, which indicates that the porous structure in MoO
3/C enhances the specific surface area and effectively shortens the diffusion lengths of charge carriers, thus facilitating the insert/extraction process of Li ions.
The cycle performance of both MoO
3/C and pristine MoO
3 at a current density of 0.5 A/g (the initial cycle current density is 0.1 A/g) in the voltage range of 0.01 - 3 V is plotted in
Fig. 4b. The initial discharge capacities of MoO
3/C and pristine MoO
3 are found to be 1922.5 mAh/g and 1422.0 mAh/g and then decreased to 991.5 mAh/g and 406.6 mAh/g , respectively. Likewise other TMO anodes
[12,
31], the capacity loss in the second cycle is mainly due to the irreversible decomposition of electrolyte as well as the formation of solid electrolyte interface (SEI) layer. The discharge capacity for MoO
3/C is retained at 608.0 mAh/g after 100 cycles, corresponding to a 61.3% retention with respect to the second cycle. The capacity of pristine MoO
3, however, decreases rapidly from 406.6 mAh/g (2
nd cycle) to 115.0 mAh/g (100
th cycle), corresponding only to a 28.3 % retention, apparently lower than that of MoO
3/C. Obviously, the capacity of MoO
3/C has been significantly improved as compared to that of pristine MoO
3. In this regard, it has been confirmed that the deterioration of MoO
3 cycle performance is mainly due to the volume expansion during the lithiation/delithiation process
[18]. For the MoO
3/C sample, the amorphous carbon around MoO
3 “buffers” the volume expansion/contraction caused by the intercalation/deintercalation reaction, yielding a cycle performance that is much better than that of pristine MoO
3.
EIS measurements for both MoO
3/C and pristine MoO
3 were carried out (
Fig. 4d). Two compressed semicircles in high/middle frequencies and an oblique line in low frequency can be clearly observed. As previously reported
[34-
36], the Ohmic resistance (
Rs) of the sample is determined by the intercept in the real axis; the semicircle in the high frequency range reflects the resistance related to the Li ion diffusion across the electrode-electrolyte interface (
Rf); and the semicircle in the middle frequency range represents the charge transfer resistance (
Rct). The oblique line at low frequencies is attributed to the Warburg impedance, related with the ionic diffusion into the bulk electrodes. As it can be seen, the intercept and diameter of both semicircles for MoO
3/C are much less than those for pristine MoO
3, indicating that the amorphous carbon enhances the electrical conductivity of MoO
3/C as well. In
Fig. 4d, the rate performance of MoO
3/C measured at current densities from 0.2 A g
−1 to 3 A/g
−1 is plotted. The mean discharge capacity of MoO
3 /C is 1070.6 mA/g, 939.9 mA/g, 755.9 mA/g, 462.6 mA/g, and 308.8 mA/g at a current density of 0.2, 0.5, 1, 2 and 3 A g
−1, respectively. When the current density recovers to 0.2 A/g, the MoO
3/C provides a discharge capacity of 938 mA/g. In comparison with pristine MoO
3, it is clear that MoO
3/C has a better rate performance; from the EIS results, it is acceptable that the improvement in the rate performance of MoO
3/C is mainly due to the increase in conductivity resulted from the existence of amorphous carbon.
As we know, the available capacity of LIBs is strongly related to their operation temperature. Therefore, it is important to explore the electrochemical performance of MoO
3/C at different temperatures.
Fig. 5a shows the cycle performance of MoO
3/C conducted at various temperatures. The cell with MoO
3/C anode material was tested at 30 ℃ in the first 10 cycles and the last 10 cycles, and at 0 ℃, 20 ℃, 30 ℃, 40 ℃, 50 ℃, 60 ℃ from the 11
th to the 100
th cycle. The initial capacity difference is usually related to the initial condition of the electrode materials, such as the infiltration of electrolyte in active material, and the formation of SEI film. It can be seen from
Fig. 5a that the lower the temperature is, the lower the specific capacity is, defining the temperature of 30 ℃ as the optimum value (11
th-12
th cycles). The relationship between discharge capacity (12
th cycle) and test temperature is plotted in
Fig. 6. The capacity retentions of MoO
3/C are 75.2%, 67.6%, 66.3%, 38.9%, 17.0% and 6.8%, respectively, when measured from 0 ℃ to 60 ℃. The relative low retentions at higher temperatures may stem from the instability of the electrode, the decomposition of the electrolyte or both. It was concluded that LiPF
6 can react with the trace amount of H
2O (in ppm level) at elevated temperature, which results in the formation of some acids, e.g. PF
5 and HF
[37]. as a consequence, the active material of MoO
3 will be corroded by these acids, leading to the decline of electrochemical performance. The corrosion of the active material at elevated temperature should be irreversible. This is the reason why the 101
st to 110
th capacity measured at 30 ℃ are obviously different, i.e. the capacity for 101st to 110
th cycle measured after previous 100 cycles at relative low temperature (≤ 20 ℃) is significantly higher than those after previous 100 cycles at higher temperature (≥ 40 ℃).
Fig. 5.
(a) cycle performance, (b) CV curves, (c) EIS plots and equivalent circuit and (d) Z’ vs. ω−1/2 plots of MoO3/C at various temperatures.
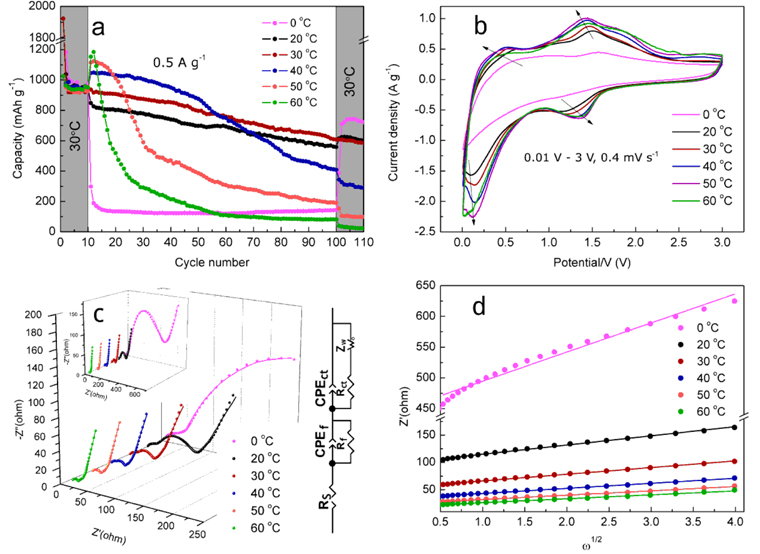
Fig. 6.
Relationship of discharge capacity, Rct, Rf and Rs with test temperature.

To further understand the effect of operation temperature on the capacity, CV and EIS were carried out at various temperatures. The experimental results are presented in
Fig. 5b and
5c. The redox peaks in CV curves measured at 0 ℃ are smaller than those at higher temperatures. This means the kinetics of redox reaction is restricted at low temperature. Furthermore, the redox peak current and the integrated area surrounded by CV curve increase gradually as increasing the test temperature, i.e. the effect of operation temperature on specific capacity is positive. As the arrows depicted in
Fig. 5b, the oxidation potentials decrease and the reduction potentials increase with the temperature rise, indicating that the electrode polarization is reduced at relatively high temperatures. In fact, the polarization details can also be acquired by EIS spectra. The Nyquist plots (
Fig. 5c) obtained at various temperatures, as well as their fitting results (
Fig. 6), reveal that there is a temperature dependence on the polarization. Generally, the kinetics of the Faradic reaction becomes sluggish as the value of
Rct increases
[38].
Fig. 6 clearly shows that the sensitivity of
Rct with respect to temperature is much stronger than that of
Rs and
Rf. And the value of
Rct is also much larger than that of
Rs and
Rf. Therefore, the influence of resistance with the change of temperature in the cell equivalent circuit arises mainly from
Rct
Furthermore, the Li ion diffusion rate is another key factor that influences the electrochemical performance of MoO
3/C. The EIS result in low frequency region which is related to Warburg impedance (
ZW) was employed to obtain the value of
DLi. The value of
ZW can be evaluated by following equation
[39]:
where
ω is the radial frequency,
σw is the Warburg impedance coefficient and can be derived from linear fitting of
Z’ vs.
ω−1/2 plot (
Fig. 5d). Then,
DLi can be obtained:
where
R is the gas constant (8.314 J·mol
−1·K
−1),
T is the absolute temperature (in K),
A is the surface area of the electrode,
F is the Faraday constant (96,485 C/mol),
σw is the Warburg impedance coefficient obtained from Eqn. (3).
C (
C =
∫idt/FVm ) is the molar concentration of Li ion in the electrode and
Vm is the molar volume of MoO
3 (
Vm = 30.7 cm
3/mol). The plot of ln
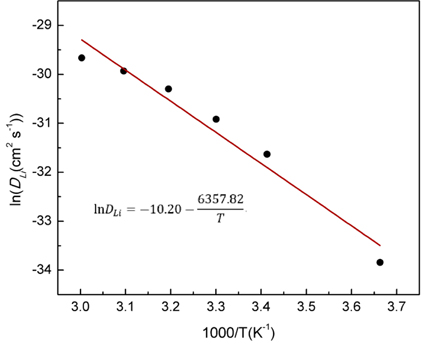
DLi vs. 1/T is exhibited in
Fig. 7. It can be seen that the temperature dependence of
DLi follows Arrhenius behavior. The modified Arrhenius equation for
DLi is
Fig. 7.
Arrhenius plot of the lithium ions diffusion coefficient of MoO3/C.
where
D0 is the pre-exponential factor,
Ea is the activation energy,
R is the gas constant,
T is the absolute temperature. Obviously, the relationship between ln
DLi and 1/T is linear and the activation energy can be obtained from the slope of the fitting line. The value of
Ea of MoO
3/C is about 52.86 kJ/mol. It is confirmed that the electrochemical lithiation reaction rate for conversion-type anode materials is mainly controlled by the diffusion of Li ion
[36]. From
Fig. 7, it can be seen that the value of ln
DLi decreases as decreasing temperature. Thus it can be concluded that the effect of temperature on the electrochemical lithiation reaction rate is positive. Therefore, it can be well understood that the polarization is aggravated by the reduction in
DLi and the increase of
Rct as decreasing temperature, corresponding to a poor electrochemical performance.