1. Introduction
Electrochemical methods are categorized in two types according to the electric signal types. One is the DC method using constant or sweeping signals of potential or current and the other one is the AC method using AC wave signals. They have pros and cons over each other. DC methods are is intuitive, easy and simple while AC methods are more sophisticated but provide richer information which DC methods cannot. Therefore, both methods are utilized in compromised ways, or sometimes complementary.
Among the AC methods, electrochemical impedance spectroscopy (EIS) is most widely used because it is a powerful electrochemical technique which can resolve a complex electrochemical observation into its elementary processes [1]. For example, when an electrochemical system is activated by electric potential, an electrochemical current is generated as an overall result mixed of electron transfer, mass transfer, charge accumulation and interactions of those processes. While DC methods such as chronoamperometry and linear sweep voltammetry can only measure the all-inclusive monotonic cur rents, electrochemical impedance spectroscopy can separate and analyze the mixed processes responding differently to frequencies [2]. Y. Zhang et al. presented a method for reconstruction of relaxation time distribution using EIS and applied the method to solid oxide fuel cells [3]. J. P. Schmidt et al. also used the distribution of relaxation time for Li-ion batteries [4]. B. A. Boukamp claimed that the use of distribution function for relaxation times has advantages and disadvantages by comparing the analysis method of complex nonlinear least square [5]. M. B. Effat et al. developed the Bayesian-based deconvolution framework for the time distribution from the data of electrochemical impedance spectroscopy [6].
Another method for analyzing EIS is the equivalent circuit presentation method which depicts a complex electrochemical process as a model consisting of resistors, capacitors and other appropriate electric elements and works best with the complex nonlinear least square fitting [7]. Then, resistance of the resistors can explain rates of mass, charge and electron transfers, and capacitance of the capacitors can evaluate the charging properties. Inductance is also used to describe properties such as releasing charges or electric energy. However, the circuit modeling method has limitations because the general electric elements are only equivalent to the ideal electrochemical processes. No electric component could represent the diffusion of molecules, so the Warburg impedance was created. The electric double layer is not perfectly capacitive, therefore a constant phase element has been practically used instead of a capacitor in many cases [8].
The constant phase element (CPE) is a variation of a capacitor. It was invented to describe impedance observed only in electrochemical systems. The impedance of the CPE, ZCPE, is expressed below as a function of ω, the angular frequency [9].
where n and Y0 are parameters characterizing the CPE.
A CPE almost inherits properties from a capacitor, but a slight difference in phase shift and magnitude responding to AC signals distinguishes the CPE from a capacitor. The degree of the phase shift of a capacitor is π/2 while that of a CPE is nπ/2, where n ranges from 0 to 1 (practically 0.8 to 1). These phase shifts are also related to the magnitude values of a capacitor and a CPE which are refereed to capacitance and Y0, respectively. When n = 1, the CPE is an ideal capacitor and the Y0 presents its capacitance. Otherwise, the CPE deviates from a capacitor and the Y0 does not directly present the capacitance any longer.
The origin of the deviation from the ideal capacitive property for the CPE is not exactly discovered but it is admitted that the practical capacitive property is well described by the CPE rather than a capacitor [8]. Over the advantage of using the CPE, a problem is faced when the physical meaning of the CPE is studied. For example, when a capacitance of an energy storage system is of interest, the deviation above should be considered: Y0 is not exactly the same as capacitance. To estimate the capacitance of a CPE, there have been several methods suggested [10–13]. C. H. Hsu et al. used a characteristic frequency at which the imaginary part is the maximum in order to find the effective capacitance and B.-Y. Chang found the effective capacitance by projecting the depressed semi-circle onto part of a perfect semi-circle. Both methods have limitations that the CPE is parallel with the resistor to make a semi-circle on the Nyquist plot. On the other hand, for a CPE with resistors in series and parallel, B. Hirschorn et al. used the equivalent time constant to derive an equation for the effective capacitance below,
There are more reports where the equivalent time constant is used to find the effective capacitance. Even though Eq. (2) is widely used in many publications, no direct evidence is provided to support the equivalence relationship of the time constants for the capacitor and the CPE. In fact, the time constant is a parameter that is measured in DC currents, however, the time constant of a CPE has not been shown in DC currents and just used in AC methods with the formula above. This evidence for Eq. (2) is important not only to confirm the relationship, but also to evaluate the electrochemical processes modeled as an equivalent circuit of a CPE and a resistor connected both in series and parallel. A typical example of such a system is a pseudocapacitor which stores electric energy as a result of faradaic reactions and it is frequently modeled as an equivalent circuit of a CPE and a resistor connected in series [14]. The CPE represents the accumulation of charges at the electrode/electrolyte interface and the resistor does the transfers of charges such as electrons, molecules, and ions through the electrolyte and at the interface. In this case, the CPE does not directly provide the capacitance from its values and needs to be calculated to result in the effective capacitance.
In this report, we show that the time constants of a capacitor and a CPE, even though they are not the same, are equivalent in DC currents, and derive an equation to find the effective capacitance of a CPE connected to a resistor in series. Here, the equation is derived based on both AC and DC techniques of electrochemical analysis: the former is electrochemical impedance spectroscopy (EIS) and the latter is chronoamperometry. EIS with the complex nonlinear least square fit evaluates the CPE and a resistor of the system of which a chronoamperometric curve results in the time constant equivalent to that of a capacitor and a resistor system. As a result, a simple equation is suggested for the conversion of the CPE to an equivalent capacitor. The equation is extended to a combination of serial and parallel resistors to generalize the CPE conversion. Those separate serial and parallel equations are unified to be applied to a general complex system composed of charge transfer and accumulation with an ohmic flow such as pseudo-capacitors.
2. Theory
When the electrified interface such as the electric double layer and a pseudo-capacitive layer behaves like an ideal capacitor, the electrochemical system is described as an equivalent circuit composed of a capacitor and a resistor in series. Upon a potential step applied, the electrochemical system produces a chronoamperometric curve decaying exponentially along the time. In the DC domain, the time constant (τ) of the exponential-decaying curve is defined by the time when the current reaches 1/e of the instant current at t = 0, and formulated as below with the components measured in the AC domain,
However, in most cases, the electrified interface does not behave like an ideal capacitor, and its non-ideal capacitive property is usually represented by a constant phase element (CPE) in the place of the capacitor. As the CPE has parameters of Y0 and n, the time constant should be a function of these parameters as expressed below,
Once the time constant is determined, it can be used to estimate the effective capacitance of the capacitor-resistor system equivalent to the CPE-resistor one which has the equivalent time constant. Thus, by combing Eq. (3) and (4), we learn that the effective capacitance can be calculated below,
As we know that τn is a measured value in the DC domain, the mathematical formula for τn should be represented by a function of time which might be derived by solving partial derivative equations for the current generated upon electric potential. However, it is impossible to obtain a closed-form solution. Instead, we can approach using numerical analysis if only we are interested in the time constant. As mentioned above, the current generated upon a potential step decays by the factor of 1/e at t = τ. Therefore, the time constant can be measured from the decayed current of the electrochemical system. For the system modeled as a circuit of a CPE and a resistor in series, its chronoamperometric curve i(t) is obtained by digital simulation which utilizes the inverse discrete Fourier transform to the impedance spectrum of the circuit.
3. Experimental
In order to find the time constants determined by precisely controlling the CPE with resistors, digital simulations are carried out by a home-made program coded in Python 3.8. For the digitized signals of Z(ω) and E(ω) in Eq. (7), suitable frequencies (ω) ranging from
1 M Δ t 1 2 Δ t
Next, the current in the frequency domain is calculated by Ohm’s law, i(ω) = E(ω)/Z(ω). Then, it is converted to i(t) by inverse discrete Fourier transform as expressed below,
where φ is the phase angle. The time constants are measured at the time when i(t) = i(0)e−1.
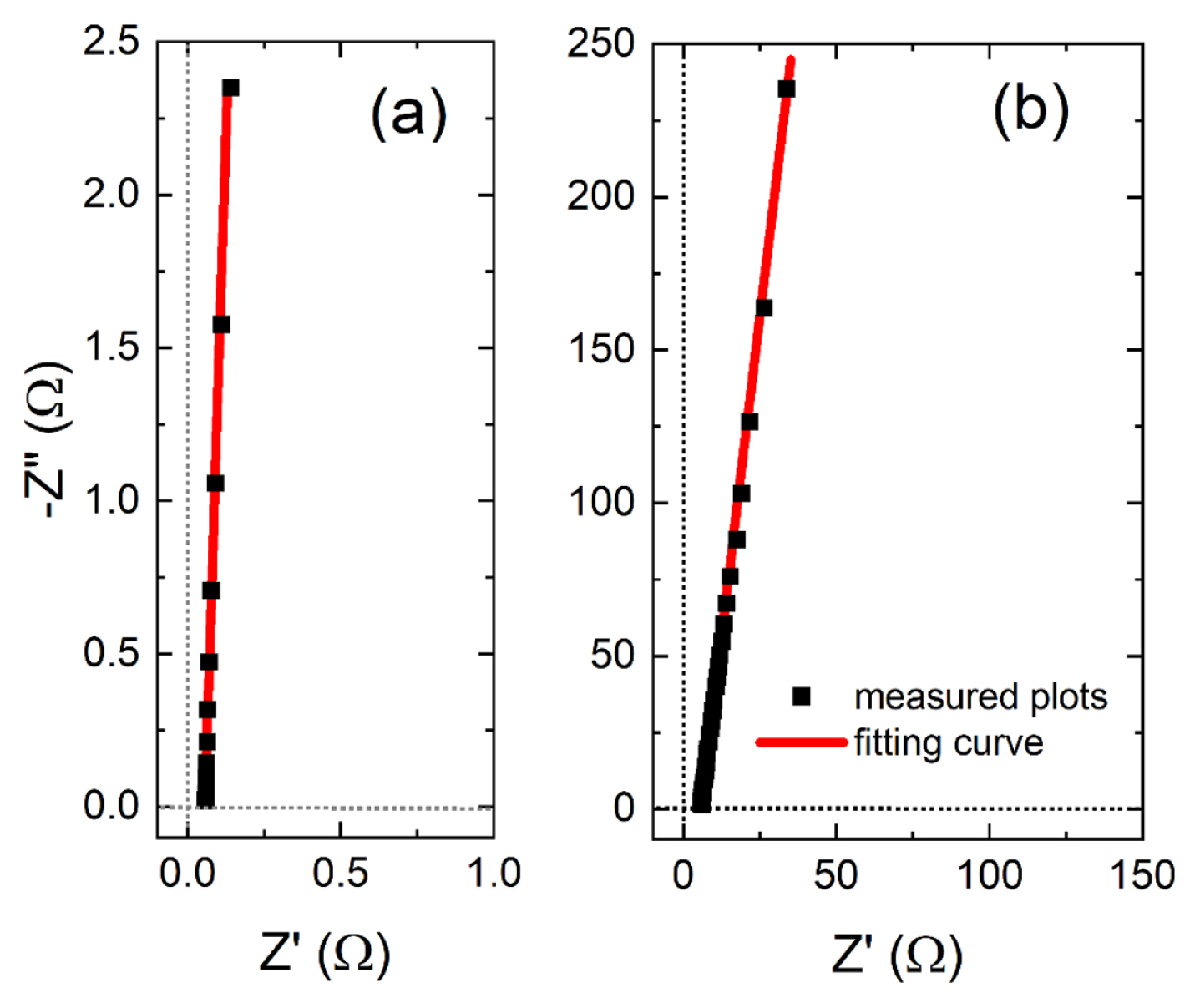
In order to confirm the theory and simulation results, a commercially available supercapacitor (BCAP0005, Maxwell Technologies Korea Co., Ltd.) and polyaniline on an ITO electrode are measured to obtain its capacitance. BCAP0005 is purchased and Polyaniline is electrochemically synthesized by applying a potential sweep from −0.2 to 1.2 V vs. Ag|AgCl at 100 mV/s in an aqueous solution containing 30 mM aniline and 0.50 M H2SO4 [15]. The electrochemical impedance spectra are acquired with SP-200 (Biologic, France). BCAP0005 is directly connected through wires and polyaniline on ITO is in 0.50 M H2SO4. The acquired data are fitted to the equivalent circuit in Fig. 1(a) using a commercial software, ZSimpWin (AMETEK, USA).
4. Results and Discussion
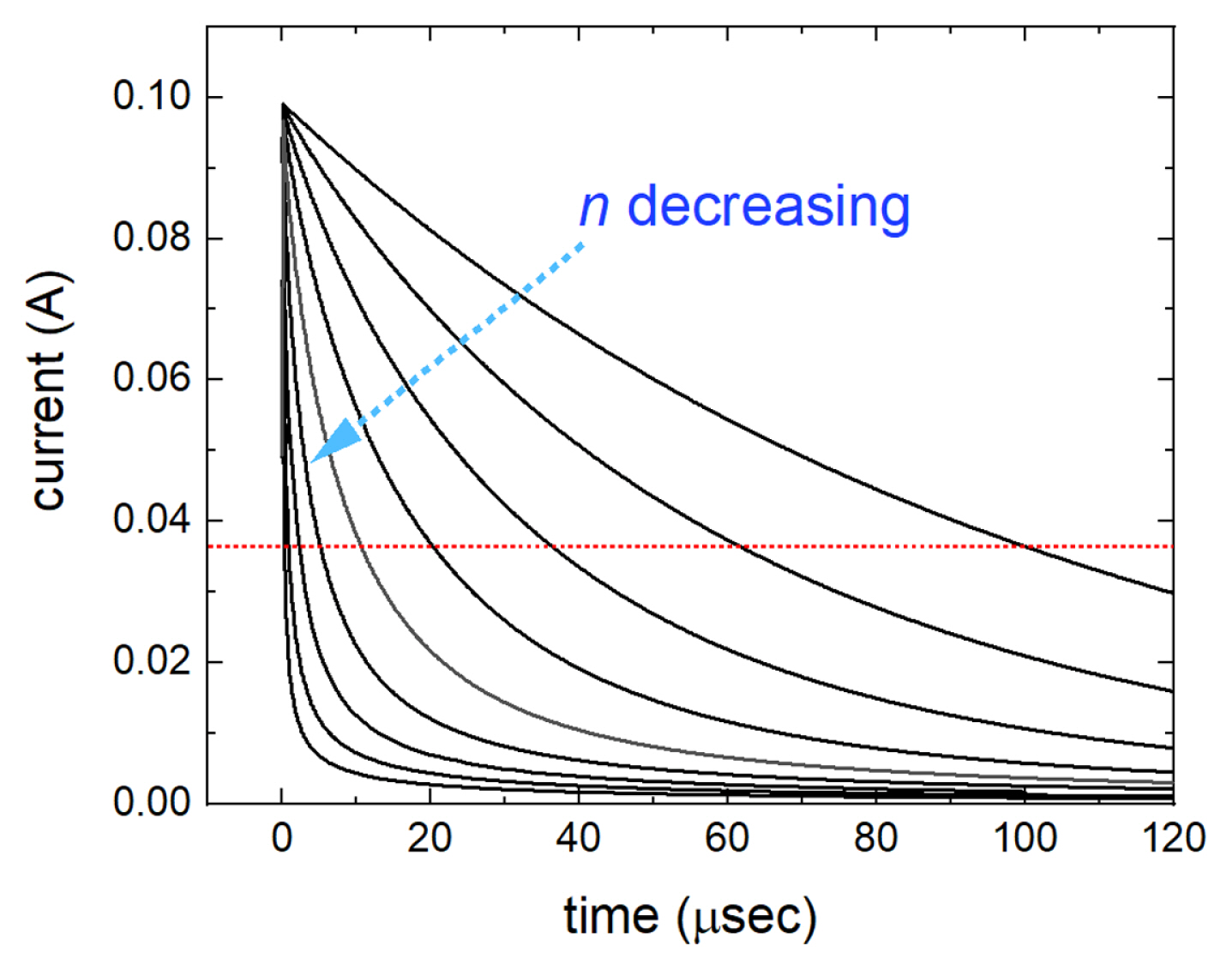
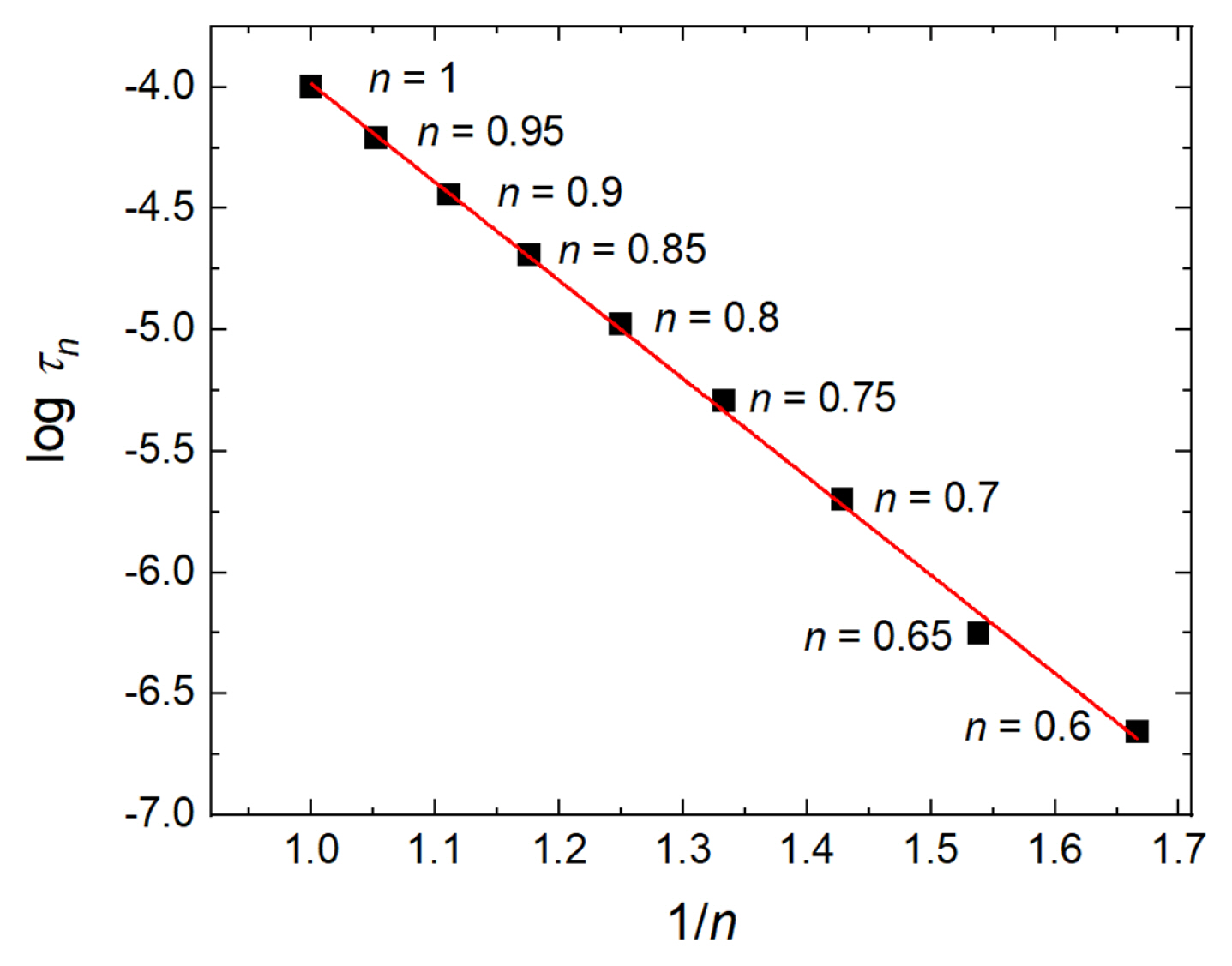
For numerical analysis, a potential step of ΔE = 1 V is made by Eq. (8) and applied on the circuit in Fig. 1(a) with parameters Rs = 10 Ω, Y0 = 10 μsn/Ω and n from 1 to 0.6. Here, n is theoretically from 0 to 1, but practically 0.8 to 1 to gain the capacitive property. Using the inverse discrete Fourier transform of Eq. (7), chronoamperometric curves are obtained and shown in Fig. 2. For n = 1, the current curve is the same as the theoretical one for a capacitor and a resistor in series, however for n < 1, the currents decay steeper as n decreases in Fig. 2. It indicates that the capacitive property is decreased with n even though Y0 is constant. Keeping in mind that the decaying current decreased by the factor of 1/e, or 0.368, the times of those currents, τn, are measured for each n value. Logarithms of the measured times are plotted against 1/n, which shows a linear relationship in Fig. 3. By varying the values of Rs and Y0, respectively, the slope of the linear line is learned to be proportional to log Rs and log Y0, respectively. Collecting the numerical analysis results, the following equation is confirmed.
We also need to notice that this equation is the same as the time constant for the circuit of a CPE and a resistor in parallel [10]. Now, Eq. (5) is defined as following,
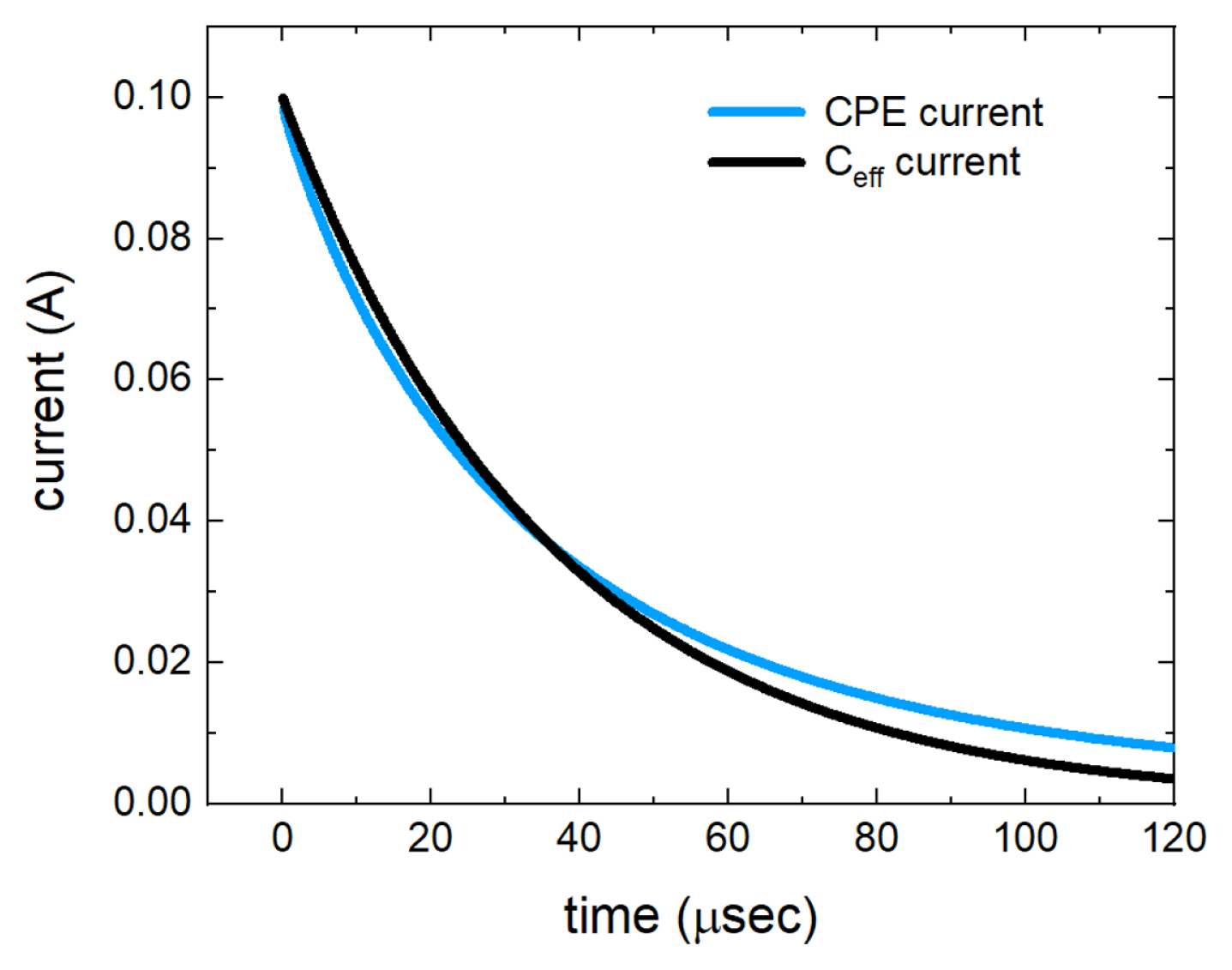
While it is obtained by the chronoamperometric approach in the DC domain, this equation is consistent with that presented by B. Hirschorn et al. which was obtained by equivalent circuit calculation in the AC domain [13]. Using this equation, the effective capacitances are calculated for the systems of which parameters are Rs = 10 Ω, Y0 = 10 μsn/Ω with different n values in Table 1. Fig. 4 compares the currents of the CPE and the equivalent capacitor when Rs = 10 Ω, Y0 = 10 μsn/Ω with n = 0.9 and Ceff = 3.61 μF. As shown in the figure, the current of the equivalent capacitor is not the same as that of the CPE, but it effectively reflects the capacitive property in terms of the relaxation time as a result of the charge accumulation process.
In order to confirm the theory, a commercially available supercapacitor (BCAP0005, Maxwell Technologies Korea Co., Ltd.) is measured SP-200 (Biologic, France) to find the capacitance. The electrochemical impedance spectrum is displayed on the Nyquist plot in Fig. 5(a). The supercapacitor, not a perfect capacitor but having high capacitance, shows a typical straight line tilted from the vertical axis. By fitting the plot to the circuit in Fig. 1(a), the values of Y0, n, and Rs are obtained 4.79 sn/Ω, 0.971 and 0.0568 Ω, respectively. The results are applied to Eq. (11) to yield the effective capacitance, 4.71 F, which is the same value as the printed capacitance of the product, 5 F (−10% to +20% tolerance). For practical application, electrochemical impedance of polyaniline is measured. Polyaniline is conducting polymer and one of the best materials for supercapacitors [16]. Once the polyaniline is electrochemically synthesized on an indium tin oxide (ITO) electrode, its electrochemical impedance spectrum is measured in 0.50 M H2SO4 solution and the Nyquist plot of the impedance is displayed in Fig. 5(b). It also shows a typical straight line of a supercapacitor and the fitting process yields Y0 = 0.0938 sn/Ω, n = 0.924, and Rs = 5.61 Ω, respectively. The effective capacitance calculated by Eq. (11) is 89 mF/cm2.
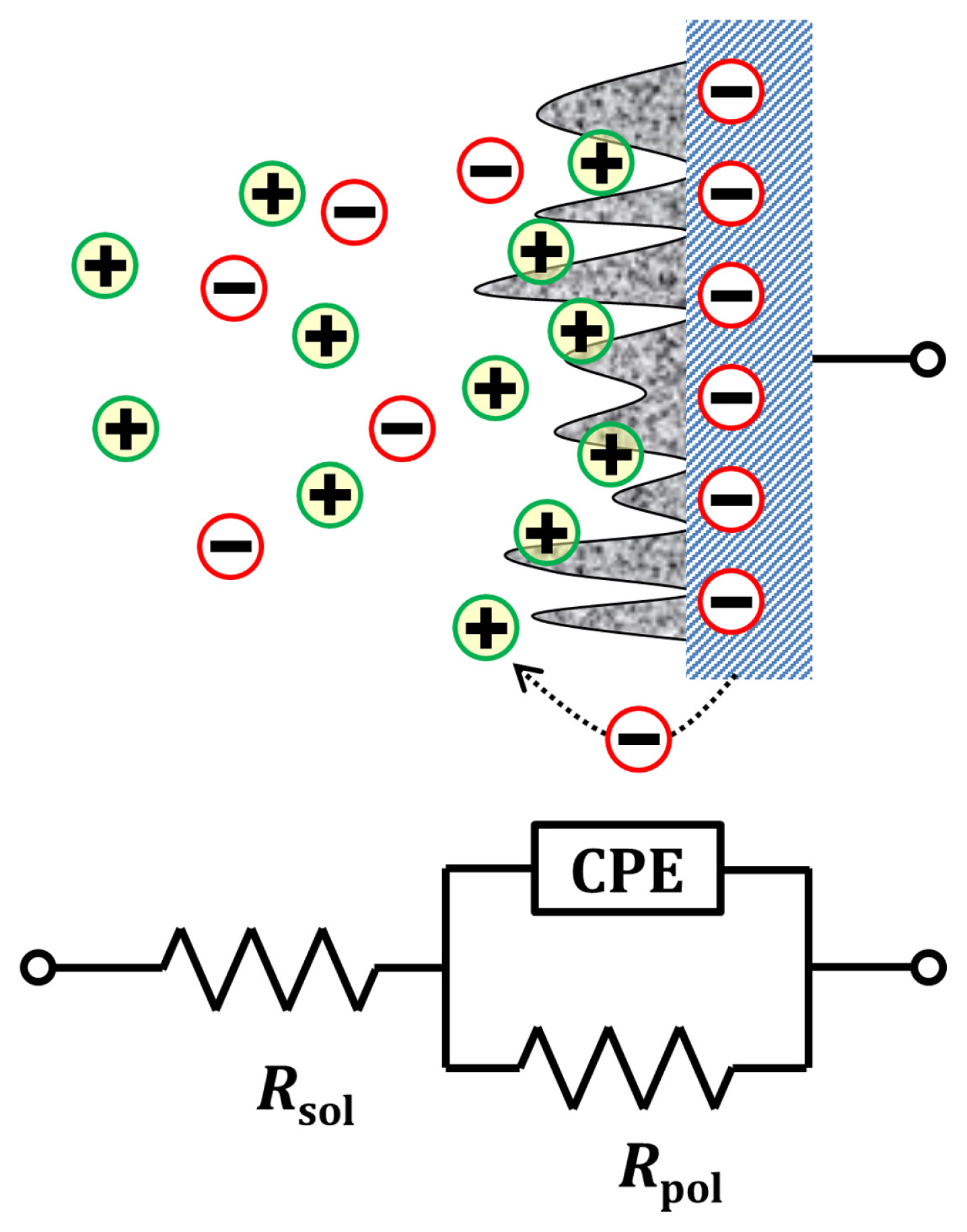
Even though the method that a CPE is converted to its equivalent capacitor based on the chronoamperometric approach is successful with a connection to a resistor either in series, it should be applicable to a system modeled as a combination of serial and parallel connections for the general purpose. The simplest equivalent circuit is shown in Fig. 6. The electrode is covered with a porous material which can accumulate charges with faradaic reaction taking place simultaneously in a conductive medium: the whole processes are described as a CPE, a polarization resistance (Rpol) in parallel and a solution resistance (Rsol) in series, respectively. According to Thevenin theorem [17] this equivalent circuit can be re-organized to the circuit in Fig. 1 by converting Rsol and Rpol to Rs and Ceff of which the resistance and the effective capacitance are following,
This equation also gives the same results that B. Hirschorn et al. presented even though the approaches are different [13]. When Rpol >> Rsol, the circuit model approaches a serial CPE-Rs model and Eq. (13) becomes Eq. (11). On the other hand, when Rpol << Rsol, the model approaches a parallel CPE-Rp model and Eq. (13) becomes the equation suggested by Mansfield [10].
It is important that the effective capacitance of a CPE is dependent on both the polarization resistance by the faradaic processes at the electrified interface and the ohmic resistance in the electrolyte [18]. Table 2 summarizes the effect of the relative resistance of Rpol and Rsol according to the n value of the CPE. It is notable that the ideal capacitor (n = 1) is not affected by the parallel Rpol, but CPEs have higher effective capacitance as Rpol increases. It is known that the imperfect capacitive property of the CPE is related to the current leakage through the dielectric material described as leakage resistance [19]. This hidden resistor provides a new pathway that charges prefer to take when the parallel Rpol is increased. Then, the increased amount of charges through the CPE cause to increase the effective capacitance.
4. Conclusions
Conversion of a constant phase element (CPE) to its equivalent capacitor is necessary when charge accumulation is more important than charge transfer at the electrode interface. There are conversion methods already reported in the literature, but they may not be suitable to high density energy storages such as supercapacitors being modeled as a double-layer-like CPE with internal resistance. In the present report, a proper conversion method is provided to find the effective capacitance of a CPE with a resistor in series describing practical supercapacitors from the aspect of time constant. While the CPE is an element in frequency domain, the time constant is measured in time domain. In order to overcome this discrepancy, the AC and the DC methods are exploited in a complementary way. As a result, a simple conversion equation is obtained. Not restricted to supercapacitors, electrochemical systems are more complex and can be modeled as a combination of serial and parallel resistors according to pathways of charge and mass transfer. In order to generalize the CPE conversion methods, an extended equation is also suggested based on the DC approach. Based on this equation, it is also learned that the effective capacitance of a CPE is dependent on the parallel charge transfer process while that of a capacitor is not.