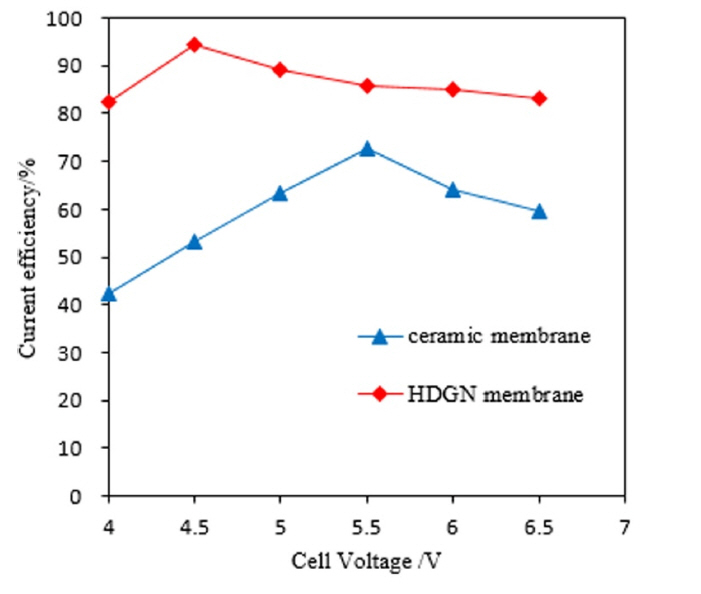
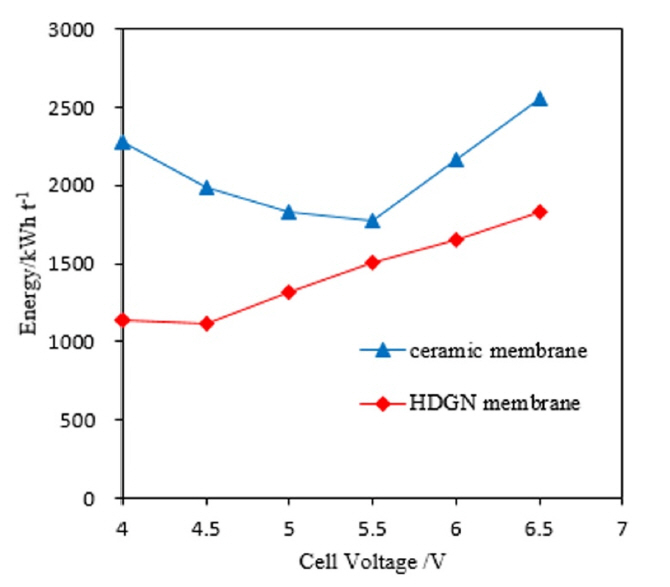
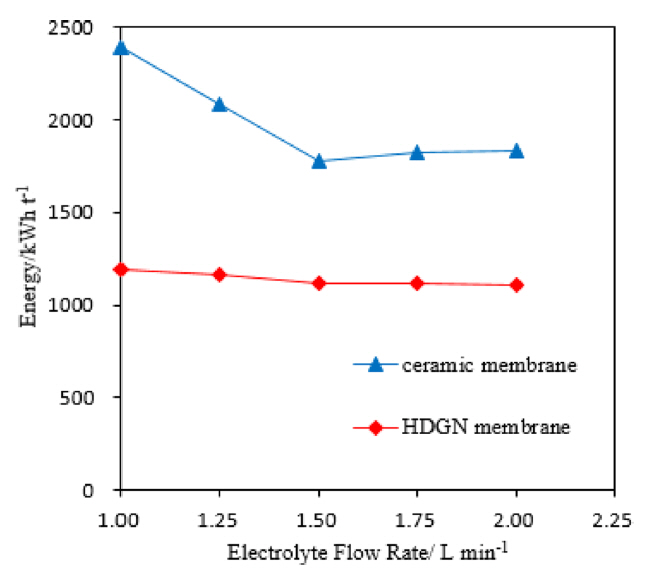
[1] SH. Lee, JM. Koo, SG. Oh and et al,
Mater Chem Phy,
2017,
194, 313–321.

[2] MK. Nizam, D. Sebastian, MI. Kairi and et al,
Sains Malay,
2017,
46, 1039–1045.

[3] LR. Bennedsen, In situ chemical oxidation: The Mechanisms and Applications of Chemical Oxidants for Remediation Purposes. Elsevier B.V., Amsterdam, 2014.
[4] N. Xu and . Guangdong, Chem Ind (Natural Science Edition), 2015, 42, 114–114.
[5] KK. Hii, K. Hellgardt, GH. Kelsall and et al, Apparatus and method for production of oxidants. WO/2016/193738A1.
[6] D. Liu, Chem Eng Des Commun, 43, 1072017.
[7] SK. Amin, MH. Roushdy, SA. El-Sherbiny and et al, Int J Appl Eng Res, 2016, 11(12), 7708–7721.
[8] JR. Davis and JC. Baygents,
J Farrell, Electrochim Acta,
2014,
44(
7), 841–848.


[9] JR. Davis and JC. Baygents,
J Farrell, Electrochim Acta,
2014,
44(
7), 841–848.


[10] VB. Martos, E. Herrero and JM. Feliu,
J Electrochim Acta,
2017,
241, 497–509.

[11] ZF. Pan, L. An, TS. Zhao and et al,
Prog in Energy Combust Sci,
2018,
66, 141–175.

[12] S. Paul,
J Electrochem Sci Technol,
2016,
7(
2), 115–131.


[13] GJ. Hwanga, SW. Kimb, DM. In and et al,
J Ind Eng Chem,
2018,
60, 360–365.

[14] H. Feng, Y. Xiong and B. Wu, J Chem Ind Eng (China), 2017, 68, 4691–4701.
[15] H. Strathmann, A. Grabowski and G. Eigenberger,
J Ind Eng Chem Res,
2013,
52(
31), 10364–10379.

[16] J. Zhao, P. Sun, L. Wang and et al, J Zhengzhou Univ (Engineering Science), 2006, 27, 109–112.
[17] H. Nan and Y. Wang, Chem Ind Eng, 2013, 30, 53–58.
[18] J. Zhu, KK. Hii and K. Hellgardt,
J Acs Sustain Chem Eng,
2016,
4, 2027–2036.

[19] C. Wang, JB. Zhou and LP. Gao,
J Electrochem Sci Technol,
2018,
9(
1), 37–41.


[20] Y. Wan and S. Xia, Water Purif Technol, 2016, 35, 102–104.
[21] H. Vogt,
Electrochim Acta,
2012,
78, 183–187.

[22] A. Li, S. Wang and X. Song, Environ Sci Manag, 2017, 42, 102–105.
[23] K. Tangphant, K. Sudaprasert and S. Channarong,
Russ J Electrochem,
2014,
50(
3), 253–259.


[24] D. Zhang, X. Song and J. Dong, Chlor-Alkali Ind, 2016, 52, 19–20.
[25] Y. Wang, Y. Xiong and J. Zhang, J Southeast Univ, 2015, 45, 738–744.