1. Introduction
Recently, Lithium ion batteries have been widely investigated due to the demands for high energy density, power and safety in large-scale applications such as PHEV, HEV, EV and energy storage systems. LiCoO2 cathode material has commercialized dominantly thus far, because it has good productivity and cycling performance. Co, however, as one of the base materials, is expensive and highly toxicity. Furthermore, LiCoO2 shows relatively low thermal stability and discharge capacity, which are essential in new cathode materials for the next generation lithium ion batteries.
Therefore, many different kinds of cathode materials have been examined extensively in the last decade such as Li
2MnO
3, LiFePO
4, LiCo
xNi1-
xO
2, LiNi
xMn
yCo
(1-x-y)O
2, etc. to replace LiCoO
2.
1-5) Among those materials, LiNi
xMn
yCo
(1-x-y)O
2, generally referred to as NCM, has been reported to enhance thermal stability and electrochemical properties by substituting transition metals (Mn, Ni or Al) in LiCoO
2 for Co.
3) Especially, the solid solution created by Li
2MnO
3 and LiMO
2 (M = Mn, Ni and Co), a complex layered compound, is considered to be promising cathode material because of its high reversible capacity of about 230-250 mAh/g when it is charged up to 4.8 V vs Li
+/Li, and its structural stability attributed to electrochemically inactive Mn
4+ ions during charge and discharge.
6-10 On the other hand, as the content of Mn ions increase, discharge capacity considerably decreases at a high current rate because of the high internal resistance in the electrode caused by the Jahn-Teller distortion, due to the Mn
3+ ions.
8,10) Therefore, one of the most crucial factors in order to improve the electrochemical properties of lithium ion batteries is to minimize the internal resistance.
As a step toward improving the electric conductivity of the 0.3Li
2MnO
3┬Ę0.7LiMn
0.55Ni
0.30Co
0.15O
2 cathode material, VGCF was chosen in this study based on the previous result as a conductive material. It was reported that VGCF connected among particles and decreased the internal resistance.
11) Therefore, it is possible to assume that VGCF could facilitate the Li ion and electron migration among particles.
In this aspect, the effects of the VGCF as a conductive material on 0.3Li2MnO3┬Ę0.7LiMn0.55Ni0.30Co0.15O2 cathode material were investigated in terms of its electrochemical performances in this study.
2. Experimental
The metal precursors of 0.3Li
2MnO
3┬Ę0.7LiMn
0.55Ni
0.30 Co
0.15O
2 cathode material were prepared via a co-precipitation method using MnSO
4┬ĘH
2O, NiSO
4┬Ę6H
2O, and CoSO
4┬Ę7H
2O as starting materials. At the same time, NaOH and NH
4OH as chelating agents were fed into the reactor in appropriate amounts and pH was maintained in the range of 11-11.5 during the process.
12) After co-precipitation, (Ni, Co, Mn)(OH)
2 precursors were filtered, washed five times by distilled water, and then dried at 100Ōäā for 24 h. Subsequently, the powder with stoichiometric LiOH┬ĘH
2O was thoroughly mixed and annealed at 900Ōäā for 3 h in air.
In order to fabricate the electrode, a cathode material, super pure black (SPB) as conductive materials, and poly vinylidene fluoride (PVDF) as a binder, were slurried with n-methyl pyrrolidone (NMP) solvent in the ratio of 84:8:8 (wt ’╝ģ). To compare the effects of VGCF, an electrode with 3 wt ’╝ģ VGCF was also produced in the ratio of 84:5:3:8 (wt ’╝ģ). The slurry after blending was coated on an aluminum current collector foil and dried at 100Ōäā for 12 h.
X-ray diffraction analysis was employed to confirm the crystal structure and impurities in the synthesized active material, using an X-pert PW3830 (Philips Co.) with Cu K╬▒ radiation under conditions of 40 kV and 30 mA. In addition, a field emission scanning electron microscope (FE-SEM, S-4800, Hitachi Co.) was used to observe the particle size and surface morphology.
For electrochemical measurements, coin-type cells (CR2032 type) consisting of a lithium foil as an anode, a separator (Celguard┬«3501), and a cathode electrode, ethylene carbonate (EC) / dimethyl ethyl carbonate (DEC) = 1/1 (vol.’╝ģ) with dissolved LiPF6 used as the electrolyte were constructed in a dry room.
The electrochemical performances were measured by a battery cycler (TOCAT-3100, TOYO System). The 0.3Li2MnO3┬Ę0.7LiMn0.55Ni0.30Co0.15O2 cathode electrode was charged and discharged in the voltage range of 2.0 - 4.6 V by the constant current (CC) - constant voltage (CV) method. Rate capability was evaluated at current rates of 0.1 C, 1 C, 2 C and 5 C while the cycling performance was tested at a current rate of 1 C at room temperature. EIS was measured for the discharged cell using an IM6 (ZAHNER, Germany) in the frequency range of 1 Hz - 1 MHz at 10 mV of amplitude, and cyclic voltammogram (CV) was operated at 0.1 mV/s between 2.0 V and 4.6 V at room temperature.
3. Results and discussion
The XRD pattern of the co-precipitated 0.3Li
2Mn O
3┬Ę0.7LiMn
0.55 Ni
0.30Co
0.15O
2 cathode material was shown in
Fig. 1. The pattern measured in this study was found to well coincide with typically hexagonal ╬▒- NaFeO
2 structure and previously reported results as indexed. In addition, (018) and (110) reflections were clearly split and a Li
2MnO
3-like region around 21-23┬░ was observed, indicating that the complex layered structure between Li
2MnO
3 and LiMn
0.55Ni
0.30Co
0.15O
2 were well formed without any remarkable impurities.
7)
Fig.┬Ā1.
XRD pattern of the 0.3Li2MnO3┬Ę0.7LiMn0.55Ni0.30 Co0.15O2 powder.
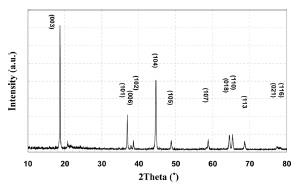
Fig. 2 showed the morphology of the 0.3Li
2Mn O
3┬Ę0.7LiMn
0.55Ni
0.30Co
0.15O
2 electrode together with its constituent materials observed using the FE-SEM. The average particle size of the cathode material synthesized in this study was approximately 300-500 nm.
Fig. 2(a)-(c) represent the 0.3Li
2Mn O
3┬Ę0.7LiMn
0.55Ni
0.30Co
0.15O
2 powder, SPB and VGCF, respectively. As presented in
Fig. 2(e), VGCF was considered to play the role of the connector between cathode material particles other than the pristine electrode as shown in
Fig. 2(d). In other words, VGCF could be expected to improve electric conductivity with other conductive materials, SPB in this study, by providing a path to electrically charged Li ions or electrons.
Fig.┬Ā2.
FE-SEM images of the 0.3Li2MnO3┬Ę0.7LiMn0.55Ni0.30 Co0.15O2 electrode together with its constituent materials : (a) active material (b) SPB (c) VGCF (d) pristine electrode (e) electrode with 3 wt’╝ģ VGCF addition.

Fig. 3 provided CV results of the 0.3Li
2Mn O
3┬Ę0.7LiMn
0.55Ni
0.30Co
0.15O
2 electrode to inspect the reversibility of redox reaction according to the addition of VGCF using fresh samples in the potential range of 2.0-4.6 V. The VGCF made no noticeable difference and no abnormal reactions were observed. The redox peaks around 3.7-4.2 V were attributed to Ni
2+/Ni
4+ followed by Co
3+/Co
4+ reactions at a higher potential region. Moreover, the decomposition of Li
2MnO
3 to Li
2O and MnO
2 was also found at 4.6 V.
Fig.┬Ā3.
Cyclic voltammograms of the Li/0.3Li2MnO3┬Ę0.7Li Mn0.55Ni0.30Co0.15O2 : (a) pristine (b) electrode with 3 wt’╝ģ VGCF addition.
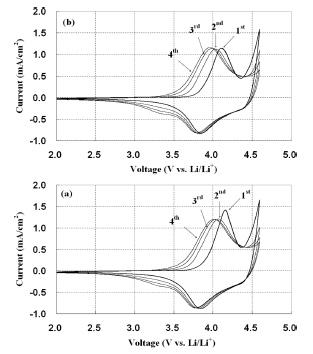
The voltage profiles of the 0.3Li
2MnO
3┬Ę0.7Li Mn
0.55Ni
0.30Co
0.15O
2 electrode after the first and second charge/discharge cycle were presented in
Fig. 4. The initial discharge capacity of electrode with VGCF was nearly 232 mAh/g, compared with 224 mAh/g of the pristine electrode at a current rate of 0.1 C. In the columbic efficiency, 75.2 ’╝ģ and 73.2’╝ģ were obtained for the electrode with VGCF and the pristine respectively, which resulted from irreversible capacity loss mainly due to the removal of Li
2O from the Li
2MnO
3 structure. As a result, the discharge capacity was somewhat improved with the VGCF, but the redox reaction region by metal ions did not change much with CV data as described in
Fig. 3.
Fig.┬Ā4.
Charge/discharge voltage profiles of the Li/0.3Li2Mn O3┬Ę0.7LiMn0.55Ni0.30Co0.15O2 cells : (a) pristine (b) 3 wt’╝ģ VGCF addition.
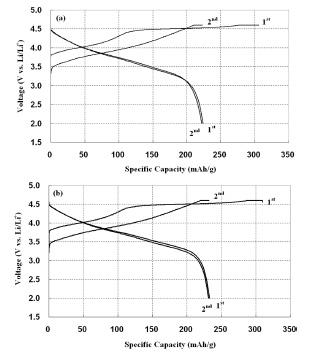
Fig. 5 represented the rate capability of a 0.3Li
2MnO
3 ┬Ę0.7LiMn
0.55Ni
0.30Co
0.15O
2 electrode at current rates of 0.1 C, 1 C, 2 C and 5 C at room temperature. At a low current density of 0.1 C, both electrodes delivered a similar discharge capacity. With an increase in current rate from 1 C to 5 C, however, discharge capacity of the pristine electrode dramatically faded from 179 to 112 mAh/g in contrast to the electrode with VGCF down from 186 to 146 mAh/g. As with the rate capability, the impedance measured in the frequency range of 1 Hz - 1 MHz showed a similar tendency, as shown in
Fig. 6. The internal resistance of the electrode with VGCF was 9.4 ╬®, lower than the pristine, 21.5 ╬®. Even after 5
th and 30
th charge/ discharge cycling, the internal resistance did not increased much for the electrode with VGCF, whereas the pristine electrode increased up to 46.3 ╬®. Thus, by adding the VGCF as a conductive substance, migration of Li ions could be facilitated through the surfaces of particles when it was charged and discharged in an electrode, resulting in the decreased internal resistance.
Fig.┬Ā5.
Rate capability of the Li/0.3Li2MnO3┬Ę0.7LiMn0.55 Ni0.30Co0.15O2 cells at the 0.1 C, 1 C, 2 C and 5 C rate : (a) pristine (b) 3 wt’╝ģ VGCF addition.
Fig.┬Ā6.
AC Impedance spectra of the Li/0.3Li2MnO3┬Ę0.7Li Mn0.55Ni0.30Co0.15O2 cells: (a) after 2nd cycle, (b) after 5th cycle and (c) after 30th cycle.

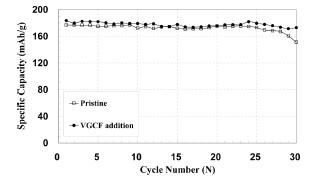
The electrode with VGCF showed a superior discharge capacity of 173 mAh/g compared to the pristine electrode of about 151 mAh/g and capacity retention of 93’╝ģ and, 85’╝ģ respectively after the 30th charge/discharge cycling at a 1 C rate in
Fig. 7. In comparison with the impedance profiles as shown in
Fig. 6, the impedance of the pristine electrode was comparable to the electrode with VGCF after 5
th cycles while the impedance of the pristine electrode significantly increased in contrast to those of electrode with VGCF after 30
th cycles. Consequently, cycle performance of the pristine electrode showed the tendency to fade dramatically after 25
th cycles.
Fig.┬Ā7.
Cycle-life performances of the Li/0.3Li2MnO3┬Ę0.7Li Mn0.55Ni0.30Co0.15O2 cells operated between 2.0 and 4.6 V at the 1 C rate : (a) pristine (b) 3 wt’╝ģ VGCF addition.
In conclusion, the VGCF enhanced not only rate capability at a high current rate, but also cycle performance by improving the electric conductivity of an electrode.
4. Summary
The 0.3Li2MnO3┬Ę0.7LiMn0.55Ni0.30Co0.15O2 cathode material was synthesized using a co-precipitation method. The traditional layered hexagonal ╬▒-NaFeO2 structure was confirmed to be without impurities by an X-ray diffraction. VGCF as a conductive material was well mixed with 0.3Li2MnO3┬Ę0.7LiMn0.55 Ni0.30Co0.15O2 particles and SPB. In electrochemical tests, the electrode with VGCF showed resistivity lower than the pristine electrode. In addition, 146 mAh/g of discharge capacity was delivered at a current rate of 5 C and 93’╝ģ of the capacity retention was also obtained in the electrode with VGCF even after the 30th charge/discharge cycling. Consequently, it was suggested in this study that VGCF as a conductive material was effective in improving electrochemical properties including rate capability, impedance, and cycle-life performance, and it provided the passage way to the cathode materials, particularly Li ions and electrons for electric conductivity.
ACKNOWLEDGEMENTS
This research was supported by a grant from the Energy Technology R&D Programs of the Ministry of Knowledge Economy, Korea (No. 2008-11-0055).
References
1.
Ding C.X.
Meng Q.S.
Wang L.
Chen C.H.
Mater. Res. Bull.,
2009,
44, 492.

2.
Jin S.J.
Song C.H.
Park K.S.
Stephan A.M.
Nahm K.S.
Lee Y.S.
Kim J.K.
Chung H.T.
J. Power Sources,
2006,
158, 620.

3.
Shaju K.M.
Subba Rao G.V.
Chowdari B.V.R.
Electrochim. Acta.,
2002,
48, 145.

4.
Kang B.W.
Ceder G.
Nature.,
2009,
458, 190.

5.
Sun Y.K.
Myung S.T.
Park B.C.
Prakash J.
Belharouak I.
Amine K.
Nature Mater.,
2009,
8, 320.

6.
Frncis A
Daniela K
Michael T
Leila Z
Judith G
Nicole L
Gil G
Boris M
Doron A
J. Electrochem. Soc., 2010, 157, 1121.
7.
Thackeray M.M.
Johnson C.S.
Vaughey J.T.
Li N.
Hackney S.A.
J. Mater. Chem.,
2005,
15, 2257.

8.
Guo X.J.
Li Y.X.
Zheng M.
Zheng J.M.
Li J.
Gong Z.L.
Yang Y.
J. Power Sources,
2008,
184, 414.

9.
Thackeray M.M.
Kang S.H.
Johnson C.S.
Vaughey S J.T.
Hackney A.
Electrochem. Commun.,
2006,
8, 1531.

10.
Johnson C.S.
Kim J.S.
Lefief C.
Li N.
Vaughey J.T.
Thackeray M.M.
Electrochem. Commun.,
2004,
6, 1085.

11.
Jin B.S.
Doh C.H.
Moon S.I.
Yun M.S.
Jeong J.K.
Nam H.D.
Park H.G.
J. Korean Electrochem. Soc., 2004, 7, 4.
12.
Lee M.H.
Kang Y.J.
Myung S.T.
Sun Y.K.
Electrochim. Acta.,
2004,
50, 939.
















